
Types of line spectra Continuous spectrum of an incandescent lamp (mid) and discrete spectrum lines of a fluorescent lamp (bottom) These "fingerprints" can be compared to the previously collected ones of atoms and molecules, and are thus used to identify the atomic and molecular components of stars and planets, which would otherwise be impossible. Spectral lines are often used to identify atoms and molecules. 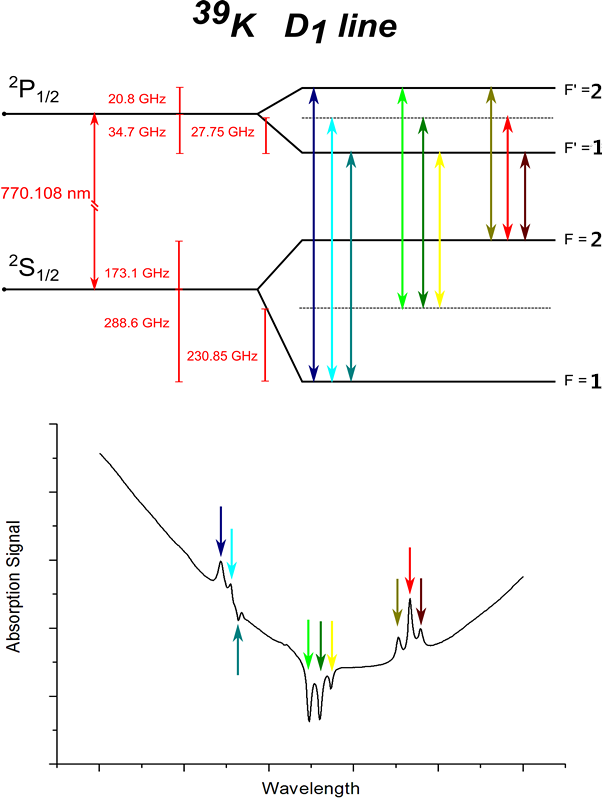
It may result from emission or absorption of light in a narrow frequency range, compared with the nearby frequencies. Here, Fraunhofer lines in sunlight and Rayleigh scattering of this sunlight is the "source." This is the spectrum of a blue sky somewhat close to the horizon, looking east with the sun to the west at around 3–4 pm on a clear day.Ī spectral line is a weaker or stronger region in an otherwise uniform and continuous spectrum. ISBN 9783709156568.Absorption spectrum with Absorption lines (discrete spectrum) Absorption lines for air, under indirect illumination, with the direct light source not visible, so that the gas is not there directly between source and detector. Struktur der Materie in Einzeldarstellungen. Linienspektren und Periodisches System der Elemente. "The Origin of the S, p, d, f Orbital Labels". Historical Studies in the Physical Sciences. "The Spectrum of ζ Puppis and the Historical Evolution of Empirical Data". The Historical Development of Quantum Theory. ^ a b Mehra, Jagdish Rechenberg, Helmut ().On the structure of the line-spectra of the chemical elements". Lines Of Light: The Sources Of Dispersive Spectroscopy, 1800-1930. "Revision of the Series in the Spectrum of Calcium". Archived from the original (PDF) on September 24, 2015. Washington: National Bureau of Standards. Atomic Transition Probabilities Volume II Sodium Through Calcium A Critical Data Compilation (PDF). "A Procedure to Obtain the Effective Nuclear Charge from the Atomic Spectrum of Sodium" (PDF). ^ a b Atomic, Molecular and Laser Physics."On a New Law Connecting the Periods of Molecular Vibrations". ^ Schuster, Arthur (31 December 1986). 
"The New Series in the Spectrum of Hydrogen". "Some Recent Discoveries in Spectrum Series". Journal of the Royal Astronomical Society of Canada. Others followed this use in the 1930s and the terminology has remained to this day. įriedrich Hund introduced the s, p, d, f notation for subshells in atoms. Heinrich Kayser, Carl Runge and Johannes Rydberg found mathematical relations between the wave numbers of emission lines of the alkali metals. In 1915 proof was given that it was actually ionised helium - helium II. This was believed to be the sharp series of hydrogen. Pickering found a new series of lines in the spectrum of ζ Puppis. Īrno Bergmann found a fourth series in infrared in 1907, and this became known as Bergmann Series or fundamental series. Rydberg continued the use of sharp and diffuse for the other lines, whereas Kayser and Runge preferred to use the term second subordinate series for the sharp series. In 1890 the lines that also appeared in the absorption spectrum were termed the principal series. They classified alkali metal spectral lines into sharp and diffuse categories. They were the first to use the term "sharp" for the lines. They noticed that lines for sodium were alternating sharp and diffuse. History Īt Cambridge University George Liveing and James Dewar set out to systematically measure spectra of elements from groups I, II and III in visible light and ultraviolet that would transmit through air. Magnesium has a sharp series of triplets and a sharp series of singlets. Calcium Ĭalcium has a sharp series of triplets and a sharp series of singlets. The sharp series of singlet lines has series letter S and formula 1P-mS. Potassium potassium sharp series Ī sharp series of triplet lines is designated by series letter s and formula 1p-ms. When n tends to infinity the diffuse and sharp series end up with the same limit. The sharp series has given the letter s to the s atomic orbital or subshell. The sharp series was important in the development of the understanding of electron shells and subshells in atoms. The lines get closer and closer together as the frequency increases never exceeding the series limit. The spectral lines include some in the visible light, and they extend into the ultraviolet. The sharp series is a series of spectral lines in the atomic emission spectrum caused when electrons descend from higher-energy s orbitals of an atom to the lowest available p orbital.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
